Recent Congress Publications
For Boehringer Ingelheim, cancer care is personal - today and for future generations. Our long-term commitment to oncology is to pursue leading science to develop innovative cancer treatments across different tumor types.
Page last updated: March 2026
ELCC 2026

A Phase I dose escalation trial of obrixtamig + ezabenlimab in patients with small cell lung cancer (SCLC) or other neuroendocrine carcinomas (NECs) expressing DLL3
Author(s): Mazières et al.
ENETS 2026

DAREON®-7: Phase I open-label dose-escalation/expansion study of first-line obrixtamig (BI 764532) plus platinum-doublet chemotherapy in patients with DLL3-positive neuroendocrine carcinomas
Author(s): Capdevila et al.
ASCO GU 2026
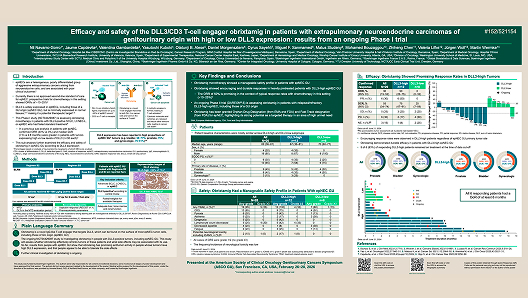
Efficacy and safety of the DLL3/CD3 T-cell engager obrixtamig in patients with extrapulmonary neuroendocrine carcinomas of genitourinary origin with high or low DLL3 expression: results from an ongoing Phase I trial
Author(s): Navarro-Gorro et al.
NANETS 2025
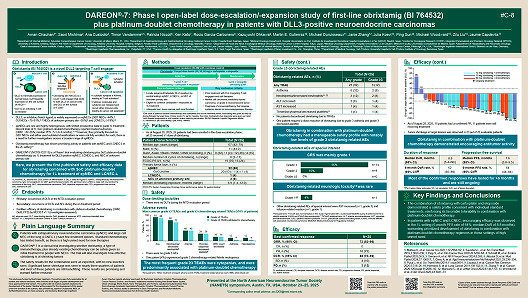
DAREON®-7: Phase I open-label dose-escalation/-expansion study of first-line obrixtamig (BI 764532) plus platinum-doublet chemotherapy in patients with DLL3-positive neuroendocrine carcinomas
Author(s): Chauhan et al.
ESMO 2025

Ezabenlimab (BI 754091) and induction mDCF (modified docetaxel, cisplatin, and 5-fluorouracil) followed by adaptive chemoradiotherapy in patients with stage III squamous cell anal carcinoma: final results of the Phase II INTERACT-ION study
Author(s): Kim et al.

An open-label, Phase Ib trial to assess the safety and efficacy of SIRPα monoclonal antibody BI 770371 in combination with pembrolizumab with or without cetuximab compared with pembrolizumab monotherapy for the first-line treatment of patients with head and neck squamous cell carcinoma (NCT06806852)
Author(s): Laban et al.

Phase Ia results for systemic STING agonist BI 1703880 plus ezabenlimab in patients with advanced solid tumors: progress to date
Author(s): Parkes et al.

Obrixtamig (BI 764532) in patients with relapsed/refractory DLL3-high expressing extrapulmonary neuroendocrine carcinoma: trial in progress of the dose expansion part of the Phase II DAREON®-5 trial
Author(s): Pavel et al.

DAREON®-8: A Phase I trial of first-line obrixtamig plus chemotherapy and atezolizumab in extensive-stage small cell lung carcinoma (ES-SCLC)
Author(s): Peters et al.
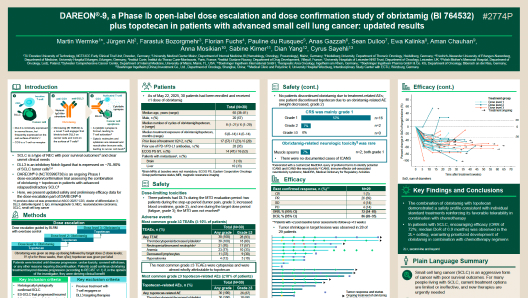
DAREON®-9, a Phase Ib open-label dose escalation and dose confirmation study of obrixtamig (BI 764532) plus topotecan in patients with advanced small cell lung cancer: updated results
Author(s): Wermke et al.
ASCO 2025

Efficacy and safety of the DLL3/CD3 T-cell engager obrixtamig in patients with extrapulmonary neuroendocrine carcinomas with high or low DLL3 expression: results from an ongoing Phase I trial
Author(s): Capdevila et al.
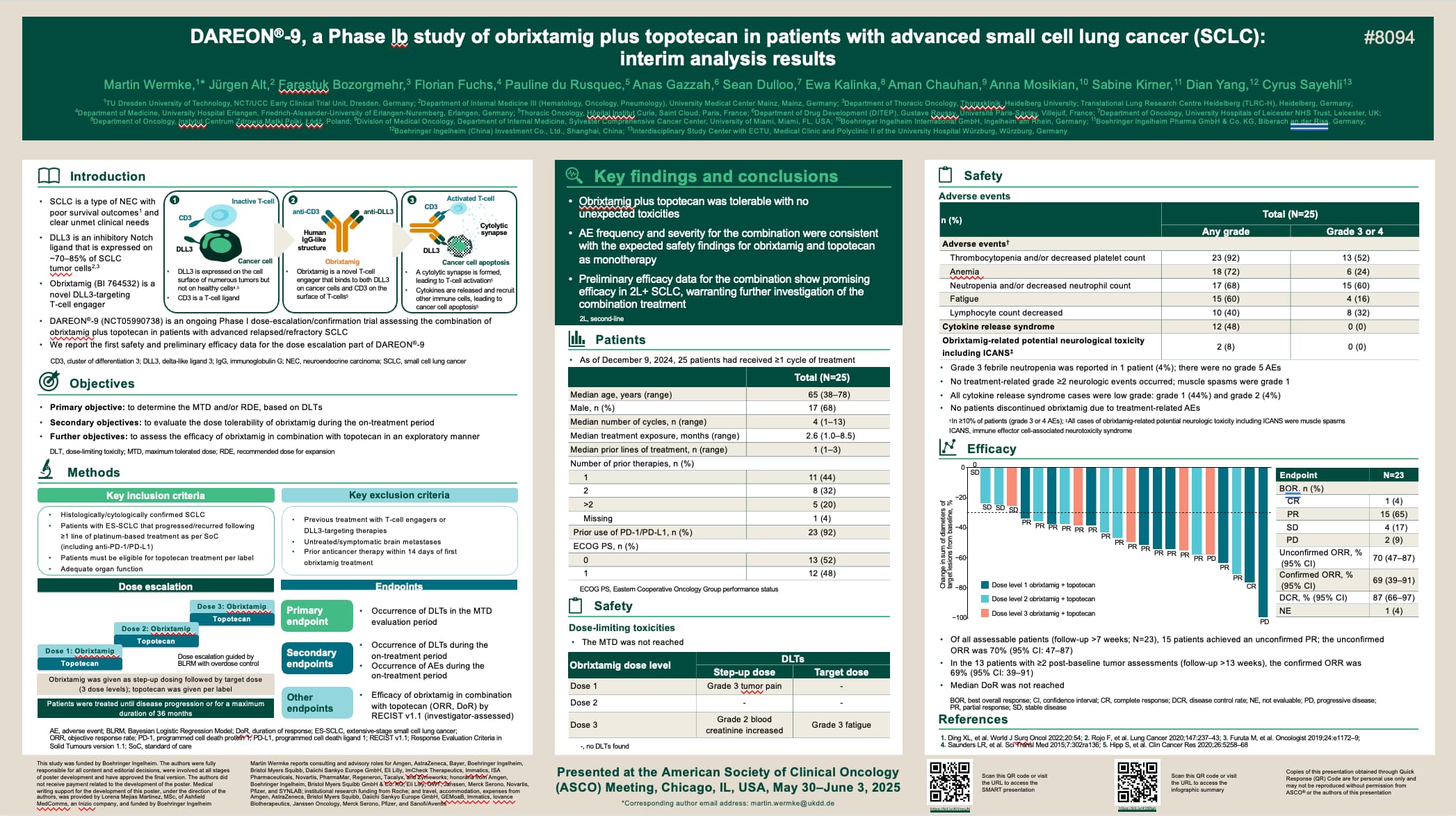
DAREON®-9, a Phase Ib study of obrixtamig plus topotecan in patients with advanced small cell lung cancer (SCLC): interim analysis results
Author(s): Wermke et al.

An open-label, Phase I trial of the SIRPα monoclonal antibody, BI 770371, alone and in combination with the PD-1 inhibitor ezabenlimab in patients with advanced solid tumors
Author(s): Wang et al.
An open-label, Phase Ib trial of the SIRPα inhibitor BI 765063 in combination with the PD-1 inhibitor ezabenlimab and cetuximab in patients with head and neck squamous cell carcinoma
Author(s): Rojas et al.

An open-label, Phase Ib dose-expansion study to assess the efficacy of CD137 FAP agonist BI 765179 plus pembrolizumab as first-line treatment in metastatic or incurable, recurrent programmed cell death ligand-1 (PD-L1)–positive head and neck squamous cell carcinoma (HNSCC)
Author(s): Shroff et al.
AACR 2025

An open-label, Phase Ib, dose-expansion study to assess the efficacy of CD137 FAP agonist BI 765179 plus pembrolizumab as a first-line treatment in metastatic or incurable, recurrent, programmed cell death ligand-1 (PD-L1)-positive, head and neck squamous cell carcinoma (HNSCC)
Author(s): Shroff et al.
